Materials
- Silver nitrate
- Lithium chloride
- Beaker
- Water
Procedure
- Place lithium nitrate on balance and measure
- Place lithium chloride in beaker and add water
- Stir beaker until lithium chloride is dissolved in water
- Place 5 drops of silver nitrate into beaker
Observations
- white cloudy solid formed
- solid swirled around in beaker
- reaction occurred very quickly
Conclusion
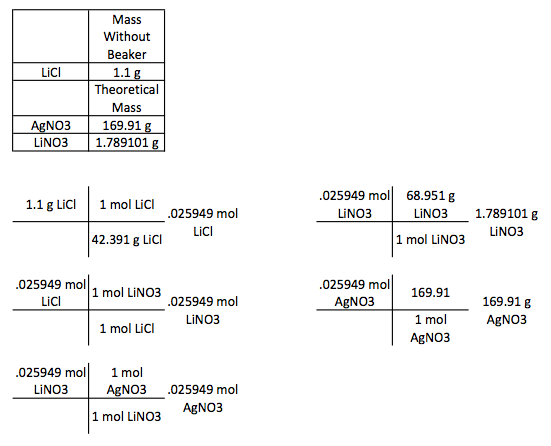
When you add silver nitrate to lithium chloride, you get lithium nitrate. There was a color change and the production of a solid. These are some signs that a chemical reaction occurred. The solid produced was AgCl and the LiNO3 was in an aqueous state. The silver chloride that was produced swirled around the beaker after the reactants were combined. The sources of error would be that all of the lithium nitrate was not absorbed in the water. Another source of error would be that I didn't measure the amount of silver nitrate was added to lithium chloride, and I didn't measure the final mass of the products. These measurements could be compared to the theoretical masses to see the percent error in my experiment.